Inhaled particles smaller than 2.5 µm ("PM2.5") are taken deep into the lungs and may be deposited there. Chronic exposure to these small particles has been linked to
The U.S. Environmental Protection Agency (EPA) has established an air quality standard of an annual average of no more than 15 µg of these particles in a cubic meter of air. (Los Angeles averaged 20 µg/m3 in 1999 and 2000.)
A survey of 217 counties in the U.S. showed an association between a reduction of 10 µg PM2.5 with an increase in life expectancy of almost 1 year. Conversely, studies in Europe showed that an increase of 10 µg PM2.5 was associated with a decrease in life expectancy of about 1 year.
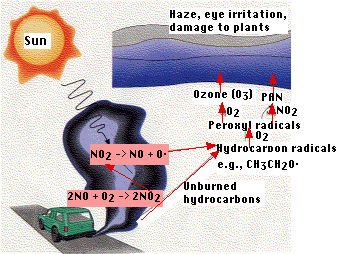 In bright sunlight
In bright sunlight
These secondary pollutants are damaging to plant life and lead to the formation of photochemical smog. PAN is primarily responsible for the eye irritation so characteristic of this type of smog.
The figure outlines representative reactions leading to the formation of photochemical smog. Radicals are atoms or molecules with unpaired electrons. They are very reactive chemically.
The catalytic converter in automobile exhaust systems reduces air pollution by oxidizing hydrocarbons to CO2 and H2O and, to a lesser extent, converting nitrogen oxides to N2 and O2.| Welcome&Next Search |