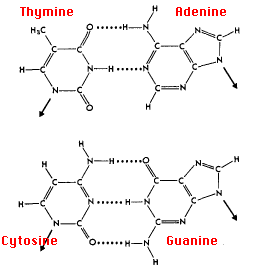
 The rules of base pairing (or nucleotide pairing) are:
The rules of base pairing (or nucleotide pairing) are:
This is consistent with there not being enough space (20 Å) for two purines to fit within the helix and too much space for two pyrimidines to get close enough to each other to form hydrogen bonds between them.
But why not A with C and G with T?
The answer: only with A & T and with C & G are there opportunities to establish hydrogen bonds (shown here as dotted lines) between them (two between A & T; three between C & G). These relationships are often called the rules of Watson-Crick base pairing, named after the two scientists who discovered their structural basis.
The rules of base pairing tell us that if we can "read" the sequence of nucleotides on one strand of DNA, we can immediately deduce the complementary sequence on the other strand.
The rules of base pairing explain the phenomenon that whatever the amount of adenine (A) in the DNA of an organism, the amount of thymine (T) is the same (called Chargaff's rule). Similarly, whatever the amount of guanine (G), the amount of cytosine (C) is the same.
| Relative Proportions (%) of Bases in DNA | ||||
|---|---|---|---|---|
| Organism | A | T | G | C |
| Human | 30.9 | 29.4 | 19.9 | 19.8 |
| Chicken | 28.8 | 29.2 | 20.5 | 21.5 |
| Grasshopper | 29.3 | 29.3 | 20.5 | 20.7 |
| Sea Urchin | 32.8 | 32.1 | 17.7 | 17.3 |
| Wheat | 27.3 | 27.1 | 22.7 | 22.8 |
| Yeast | 31.3 | 32.9 | 18.7 | 17.1 |
| E. coli | 24.7 | 23.6 | 26.0 | 25.7 |
| Welcome&Next Search |