| Index to this page |
| Discussion of antigen presentation to B cells and T cells |
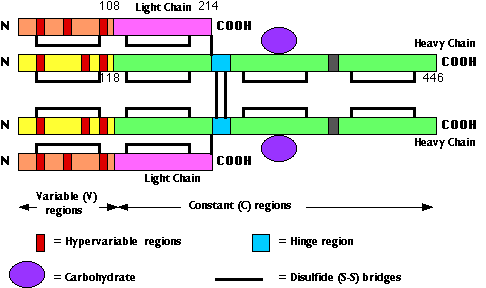
This image represents the polypeptide chain structure of a molecule of IgG. The numbers indicate the number of amino acids (counting from the amino terminal ("N"). In the actual molecule, the chains are folded to that each cysteine is brought close to the partner with which it forms a disulfide (S–S) bridge.
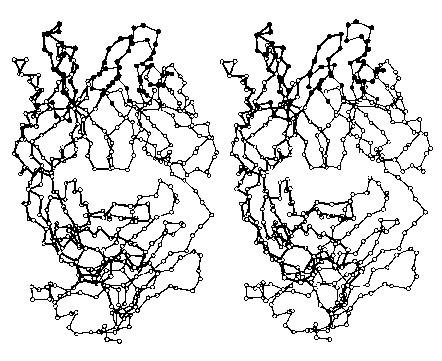
The images below (courtesy of Dr. D. R. Davies) represent the folded (tertiary) structure of an entire L chain (right side with thin connecting lines) and the V region plus the first third of the C region of a heavy chain (left side; darker lines). Each circle represents the location in 3D space of an alpha carbon. The filled circles at the top are amino acids in the hypervariable or complementary determining regions (CDRs); they form the site that binds the antigen.
Do try to fuse these two images into a stereoscopic (3D) view. I find that it works best when my eyes are about 18" (about 46 cm) from the screen, and I try to relax so that my eyes are directed at a point behind the screen.
Antibody molecules have two functions to perform:
So, V regions finger the culprit; the C regions take action.
If an antibody's H chains (see IgG above), are cut at its hinge region on the N-terminal side of the disulfide bonds holding the H chains together, 3 fragments are produced:Why 5 kinds of heavy chains? To provide for different effector functions.
| Class | H chain | L chain | Subunits | mg/ml | Notes |
|---|---|---|---|---|---|
| IgG | gamma | kappa or lambda | H2L2 | 6–13 | transferred across placenta; four subclasses: IgG1-4 in humans |
| IgM | mu | kappa or lambda | (H2L2)5 | 0.5–3 | first antibodies to appear after immunization |
| IgA | alpha | kappa or lambda | (H2L2)2 | 0.6–3 | much higher concentrations in secretions; two subclasses |
| IgD | delta | kappa or lambda | H2L2 | <0.14 | function uncertain |
| IgE | epsilon | kappa or lambda | H2L2 | <0.0004 | binds to basophils and mast cells sensitizing them for certain allergic reactions |
The subclasses of IgG and IgA are encoded by different C-region gene segments.
| If an antibody-secreting cell becomes cancerous, it will grow into a clone secreting its single class of molecule. The disease is called multiple myeloma. See how IgG, IgM, and IgA molecules appear in the serum of these patients. |
The antibodies produced by normal B cells have two identical Fab fragments with identical antigen-binding specificities. However, in the laboratory it is possible through cell fusion and genetic engineering techniques to make B cell clones that generate antibodies with differing specificities; that is, one Fab that binds a particular epitope, with the other Fab binding a different epitope.
Example: mosunetuzumab. The antibodies secreted by this B-cell clone have
Presumably, mosunetuzumab binds and activates the T cell while linking it to the malignant myeloma cell which it can then destroy (e.g., by releasing granules of granzyme B and perforin).
Mosunetuzumab is one of several bispecific antibodies that have produced substantial remissions in patients with multiple myeloma and patients with B-cell lymphomas.
The antigen receptor on most T cells is made up of two transmembrane polypeptides designated alpha and beta (thus forming a heterodimer).
Like antibodiesA small percentage of the T cells in the blood use a TCR consisting of a heterodimer of two other types of transmembrane polypeptides: gamma and delta. The function of this subset of T cells is still a mystery. [More]
Each chain of a BCR or TCR is encoded by a gene that is assembled from separate gene segments during the differentiation of the cell. The resulting gene is transcribed into a mRNA to be translated into one chain of the receptor. The number of gene segments from which the variable regions are constructed is sufficiently large that both B cells and T cells can generate more than 107 different antigen-binding sites. There is probably no epitope that could exist for which BCRs and TCRs able to bind it are not built.
| The mechanisms by which B cells and T cells generate receptors of such extraordinary diversity. |
| Welcome&Next Search |