| Index to this page |
Blood groups are created by molecules present on the surface of red blood cells (and often on other cells as well).
The ABO blood groups were the first to be discovered (in 1900) and are the most important in assuring safe blood transfusions.
The table shows the four ABO phenotypes ("blood groups") present in the human population and the genotypes that give rise to them.
| Blood Group | Antigens on RBCs | Antibodies in Serum | Genotypes |
|---|---|---|---|
| A | A | Anti-B | AA or AO |
| B | B | Anti-A | BB or BO |
| AB | A and B | Neither | AB |
| O | Neither | Anti-A and Anti-B | OO |
When red blood cells carrying one or both antigens are exposed to the corresponding antibodies, they agglutinate; that is, clump together. People usually have antibodies against those red cell antigens that they lack.
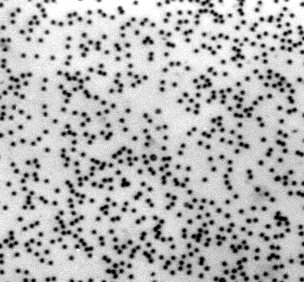
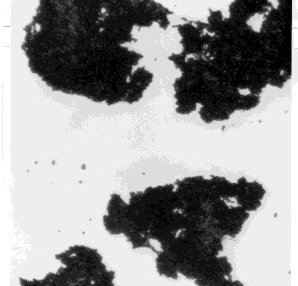
| Human red blood cells before (left) and after (right) adding serum containing anti-A antibodies. The agglutination reaction reveals the presence of the A antigen on the surface of the cells. | |
 |  |
The antigens in the ABO system are O-linked glycoproteins with their sugar residues exposed at the cell surface. The terminal sugar determines whether the antigen is A or B.
The critical principle to be followed is that transfused blood must not contain red cells that the recipient's antibodies can clump. Although theoretically it is possible to transfuse group O blood into any recipient, the antibodies in the donated plasma can damage the recipient's red cells. Thus, when possible, transfusions should be done with exactly-matched blood.
In 2007, Danish and French investigators reported the properties of two bacterial glycosidases that specifically remove the sugars responsible for the A and B antigens. This discovery raises the possibility of being able to treat A, B, or AB blood with these enzymes and thus convert the blood to Group O, the "universal donor".
Why do we have antibodies against red cell antigens that we lack? Bacteria living in our intestine, and probably some foods, express epitopes similar to those on A and B. We synthesize antibodies against these if we do not have the corresponding epitopes; that is, if our immune system sees them as "foreign" rather than "self".
Rh antigens are transmembrane proteins with loops exposed at the surface of red blood cells. They appear to be used for the transport of carbon dioxide and/or ammonia across the plasma membrane. They are named for the rhesus monkey in which they were first discovered.
There are a number of Rh antigens. Red cells that are "Rh-positive" express the one designated D. About 15% of the population have no RhD antigen and thus are "Rh-negative".
The major importance of the Rh system for human health is to avoid the danger of RhD incompatibility between mother and fetus.During birth, there is often a leakage of the baby's red blood cells into the mother's circulation. If the baby is Rh-positive (having inherited the trait from its father) and the mother Rh-negative, these red cells will cause her to develop antibodies against the RhD antigen. The antibodies, usually of the IgG class, do not cause any problems for that child, but can cross the placenta and attack the red cells of a subsequent Rh+ fetus. This destroys the red cells producing anemia and jaundice. The disease, called erythroblastosis fetalis or hemolytic disease of the newborn, may be so severe as to kill the fetus or even the newborn infant. It is an example of an antibody-mediated cytotoxicity disorder.
| Other examples of antibody-mediated cytotoxicity disorders. |
Although certain other red cell antigens (in addition to Rh) sometimes cause problems for a fetus, an ABO incompatibility does not. Why is an Rh incompatibility so dangerous when ABO incompatibility is not?
It turns out that most anti-A or anti-B antibodies are of the IgM class and these do not cross the placenta. In fact, an Rh−/type O mother carrying an Rh+/type A, B, or AB fetus is resistant to sensitization to the Rh antigen. Presumably her anti-A and anti-B antibodies destroy any fetal cells that enter her blood before they can elicit anti-Rh antibodies in her.
This phenomenon has led to an extremely effective preventive measure to avoid Rh sensitization. Shortly after each birth of an Rh+ baby, the mother is given an injection of anti-Rh antibodies. The preparation is called Rh immune globulin (RhIG) or Rhogam. These passively acquired antibodies destroy any fetal cells that got into her circulation before they can elicit an active immune response in her.
Rh immune globulin came into common use in the United States in 1968, and within a decade the incidence of Rh hemolytic disease became very low.
Several other blood group antigens have been identified in humans. Some examples: MN [Link], Duffy, Lewis, Kell.
They, too, may sometimes cause(The Duffy red cell antigen also serves as the receptor for entry by the malaria parasite Plasmodium vivax.)
| Welcome&Next Search |