| Index to this page |
In symbiosis, at least one member of the pair benefits from the relationship. The other member may be
Mutualistic relations between plants and fungi are very common. The fungus invades and lives in or among the cortex cells of the secondary roots. The association is called a mycorrhiza.
The fungus helps the host plant absorb inorganic nitrogen and phosphorus from the soil. Some mycorrhizal fungi also secrete antibiotics which may help protect their host from invasion by parasitic fungi and bacteria.
Many mushrooms are the spore-forming bodies of mycorrhizal fungi. The truffle [View] is often found in oak forests because the fungus that produces it establishes mycorrhiza on oak roots.| More on mycorrhizal fungi |
Endosymbiosis is a mutualistic relationship between a host and an organism living within its body or cells.
The pea aphid, Acyrthosiphon pisum, is an insect pest that sucks the juices from its host plant. However, plant sap is deficient in several essential amino acids. The pea aphid thrives nonetheless thanks to specialized cells within its body that contain the gamma proteobacterium, Buchnera aphidicola, that can live nowhere else. The genome of this obligate intracellular Gram-negative bacterium encodes a number of enzymes needed to complete the synthesis of the amino acids needed by its host.
In return, the aphid's genomeOne of the most important examples of mutualism in the overall economy of the biosphere is the endosymbiotic relationship between certain nitrogen-fixing bacteria and their legume hosts.
| Link to a discussion. |
A large body of evidence supports the view that intracellular endosymbiotic relationships gave rise to eukaryotes with their mitochondria and chloroplasts.
| Examine some of the evidence at Endosymbiosis and The Origin of Eukaryotes |
 The drawing shows the Nile crocodile opening its mouth to permit the Egyptian plover to feed on any leeches attached to its gums.
The drawing shows the Nile crocodile opening its mouth to permit the Egyptian plover to feed on any leeches attached to its gums.
Cleaning symbiosis is more common in fish.
Some examples:
Animals are parasitized by viruses, bacteria, fungi, protozoans, flatworms (tapeworms and flukes), nematodes, insects (fleas, lice), and arachnids (mites).
Plants are parasitized by viruses, bacteria, fungi, nematodes, and a few other plants.
Parasites damage their host in two major ways:
In 1859, the European rabbit was introduced into Australia for sport. With no important predator there, it multiplied explosively. The raising of sheep (another imported species) suffered badly as the rabbits competed with them for forage.
This picture (courtesy of Dunston from Black Star) gives you the idea. Having removed all forage plants, which ordinarily supply them with water as well as food, the rabbits had to drink from a pool.
In 1950, the myxoma virus was brought from Brazil and released. The epidemic that followed killed off millions of rabbits (perhaps 99.5% of the population). Green grass returned and sheep raising once again became profitable.But the rabbits were not eliminated. In fact, although small epidemics still occur, the rabbit population has recovered somewhat (although nowhere near its pre-1950 levels).
What happened?
Thanks to careful planning, we know.
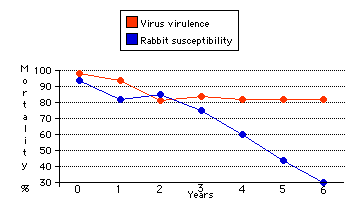
The graph (based on data of Sir Macfarlane Burnet and D. O. White) shows these mutual evolutionary adaptations over the first six years after the introduction of the virus.
Because of their growing resistance to the myxoma virus, another lethal virus — rabbit hemorrhagic disease virus (RHDV) — was introduced in the 1990s. While effective for a time, susceptibility to RHDV is diminishing just as it did for myxoma.
During the course of adapting to conditions in their host, parasites often lose structures and functions that were essential for their ancestors (and any free-living relatives).
The tapeworm has no eyes, no digestive tract, and only vestiges of nervous, excretory, and muscular systems.
While you may call them degenerate, these losses represent a gain in efficiency and improved specialization. What good would these structures be anyway in the human intestine? On the other hand, the tapeworm produces hundreds of proglottids [View]: egg-forming machines that improve the likelihood that the tapeworm will leave descendants that reach another host.
This emphasis on reproduction is also seen inM. leprae causes leprosy (Hansen's disease). It is an intracellular parasite, taking up residence in Schwann cells where, in due course, it triggers an autoimmune attack on them that leads to their destruction. The resulting loss of sensation makes it difficult to avoid injury to the extremities.
M. leprae is a mycobacterium and a close relative of M. tuberculosis, the cause of TB.
Although it was the first bacterium to be identified as a cause of human disease (in 1873), no bacteriologist has ever succeeded in cultivating it in vitro. It can, however, be propagated (slowly) in the nine-banded armadillo, and this has provided enough material to sequence its entire genome.
| M. leprae infection also occurs naturally in the wild armadillos living in a band of southern states extending from Alabama through Texas. A survey of 39 human leprosy patients in that region revealed 25 of them infected with the identical strain found in the local armadillos. |
Its sequence, which was published in the 22 February 2001 issue of Nature (Cole, S. T. et al.) — when compared to that of M. tuberculosis — provides a vivid demonstration of degeneracy at the level of the genes.
Although its genome is only about 25% smaller than that of M. tuberculosis, it has only 40% of the genes of its cousin. Many of the missing genes are still detectible, but they are now pseudogenes — genes that have mutated so that they can no longer be expressed in a protein product.
| M. tuberculosis | M. leprae | |
| Size of genome (bp) | 4,411,532 | 3,268,203 |
| Protein-coding genes | 3,959 | 1,604 |
| citrate synthase genes (for citric acid cycle) | 3 | 1 |
| pyruvate dehydrogenase genes (for citric acid cycle) | 6 | 2 |
| lactate dehydrogenase genes (cellular respiration) | 2 | 1 |
| phosphofructokinase genes (glycolysis) | 2 | 1 |
M. leprae is not an exception. The many bacterial genomes that have now been sequenced show that bacteria that are obligate intracellular parasites express far fewer proteins than bacteria that can live on culture medium.
It seems plausible that what begins as a parasitic relationship might over the course of time evolve into a mutualistic one as the two organisms evolve to minimize the damage to the host.
And there is some evidence for this. In 1966, K. W. Jeon discovered a culture of amoebas that had become infected with bacteria (60,00 to 150,000 per cell). The infection slowed their rate of growth and made them much more fragile. But five years later, the amoebas still were infected but now no ill effects could be seen. Most interesting for our question, the amoebas — or at least their nuclei — had become dependent on the bacteria.Evidently, after 5 years, the nuclei had become dependent on a bacterial function (an enzyme produced by the bacteria but no longer by the host). What started as parasitism had evolved into mutualism (the bacteria could not be grown outside their host).
But it doesn't always work like that. There are other examples where a mutualistic relationship seems to have evolved into a commensalistic or even parasitic one. Some parasitic fungi seem to have evolved from ancestors living in the mutualistic partnership of a lichen.
Some of the bacteria living in our large intestine supply us with vitamin K, thus evolving from commensalism to mutualism.
Mutually beneficial symbiotic relationships can lead to "degeneracy" also. Some marine annelid worms have completely lost the digestive tract of their relatives (like the common earthworm). One species gets its nourishment from a large population of at least 5 different species of bacteria living underneath its outer skin. The most abundant of these are chemoautotrophs (but these bacteria are gamma- and delta- proteobacteria not beta-proteobacteria) that manufacture food from carbon dioxide using the energy provided by oxidizing inorganic substances (H2S, H2) in the sediments in which the worm lives.
The nature of a symbiotic relationship can also change as circumstances change. Some fungi, bacteria, and protozoans that live harmlessly in most of us can cause opportunistic infections — that is become parasitic — in immunodeficient people, e.g., those with AIDS [Discussion].
| Welcome&Next Search |