| Index to this page |
| Link to discussion of them. |
These are encoded by an array of genes on chromosome 6 called the major histocompatibility complex (MHC).
Class I molecules consist of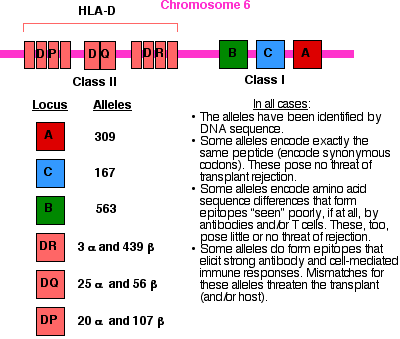
The class I transmembrane proteins are encoded by three "major" loci: HLA-A, HLA-B, and HLA-C (and also three "minor" loci: HLA-E, HLA-F, and HLA-G which will not be discussed here).
| Link to schematic of the HLA-A2 molecule (92K) |
Class I molecules are expressed at the surface of almost all the cells of the body (except for red blood cells and the cells of the central nervous system).
Class II molecules consist of two transmembrane polypeptides:The alpha and beta chains are encoded by clusters of loci in the region of chromosome 6 designated HLA-D.
Unlike class I molecules, class II molecules are expressed on only a few types of cells, chiefly antigen-presenting cells (APCs) such as dendritic cells and macrophages, as well as other cells where inflammation is occurring.
The genes of the MHC are the most polymorphic known. The graphic above shows the latest counts of alleles found at each locus in the human population. Of course, any one human can inherit a maximum of two alleles at each locus.
The diversity of alleles in the population makes possible thousands of different combinations. In a study of 1000 blood and organ donors in San Francisco that were typed for HLA-A and HLA-B,The extreme polymorphism of the MHC did not evolve to frustrate transplant surgeons and their patients.
Why, then?
The function of class I and class II molecules is to "present" antigenic peptides to the T cells of the immune system. The peptides — usually about 9 amino acids long — are bound by noncovalent forces in a groove [View] at the surface of the MHC molecule. These peptides can include fragments of protein antigens derived from intracellular pathogens (e.g. viruses).
| Discussion of antigen presentation by class I and class II molecules. |
One piece of evidence:
People infected with the human immunodeficiency virus (HIV-1) who have one particular HLA-B molecule are more resistant to developing a full-blown case of AIDS than those with other HLA-B molecules (even though some of these differ by only a single amino acid).
Another piece of evidence:
Wegner and colleagues reported in the 5 September 2003 issue of Science the results of a direct test of this hypothesis. They exposed groups of sticklebacks — differing in the number of their class II alleles — to three types of parasite (all at once). They used two species of parasitic nematode and one species of trematode). Those fish with 5–6 different alleles resisted infection better than those with fewer (or more).
So it may well be that the great diversity of class I and class II alleles in the human population has helped ensure that no single pathogen can wipe out the entire population.
Most tissue typing is done using serological methods: antibodies specific for those HLA antigens that have been identified in the human population. A reaction between cells of the subject and, for example, anti-HLA-A28 antibodies and HLA-A9 antibodies — but no other antibodies — establishes the phenotype.
At the present time, routine typing is limited to establishing the phenotype at HLA-A, HLA-B, and HLA-DR.
Coming into wider use is DNA typing, especially for HLA-D antigens. It promises to improve the sensitivity and specificity of tissue typing. The totals of numbers of alleles at each HLA locus given in the graphic above are based on DNA typing.
So what hope do these data hold for the dialysis patient awaiting a kidney transplant?
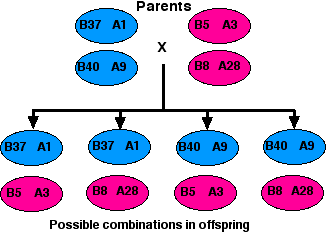
If the patient has a large family of willing donors, the odds for a good match are not bad because of the tight linkage of the HLA loci.
Assuming that no crossing over occurs within the HLA region of either the mother's or the father's two number 6 chromosomes, there are four possible combinations in which they may transmit their alleles to their children. So even if the parents carry different alleles at each locus (which is often the case), there is a 1:4 probability that any one of their children will be an exact match with any other. (Only the HLA-A and HLA-B antigens are shown here, but the tight linkage of the entire HLA region makes it likely that all the loci on each chromosome will be passed on as a block.)
But,| Number of HLA mismatches |
% kidneys surviving after 5 years |
|---|---|
| 0 | 68 |
| 1 | 61 |
| 2 | 61 |
| 3 | 58 |
| 4 | 58 |
| 5 | 57 |
| 6 | 56 |
Allografts that contain T cells of the donor can cause graft-versus-host disease (GVHD). The T cells in the transplant see the host as "foreign" and proceed to mount a widespread attack against the tissues of the host.
GVHD is an especially serious problem with grafts of bone marrow (the source of all blood cells) and cord blood. Even when the donor and recipient have identical HLA alleles, grafts of bone marrow often cause GVHD because of differences in their minor histocompatibility antigens.
Some cancer patients are now deliberately treated so vigorously with radiation and chemotherapy that their bone marrow is destroyed along with their cancer cells. In order to survive, these patients must be given stem cells to repopulate their marrow after their therapy.
In mice, GVHD can be minimized by injecting large numbers of regulatory T cells, but for humans, control of GVHD — like control of graft rejection — still depends on the use of immunosuppression.
Immunosuppression is the treatment of the patient with agents that inhibit the immune response. Presently the major players are:
These are relatives of the purines used in DNA synthesis. Because they interfere with DNA synthesis, they interfere with the rapid cell proliferation needed for immune responses. Azathioprine (trade name = Imuran) is a widely-used purine analog.
Unfortunately, these drugs also interfere with the many other tissues that depend on rapid cell division (e.g., lining of the intestine, hair follicles) so they have many unpleasant side effects. Therefore, the search for agents that specifically target immune cells goes on.
These are natural products isolated from microbial cultures. They inhibit the signaling pathway used by T cells to turn on their genes for activation, e.g., for IL-2 secretion.
Rapamycin is also known as sirolimus and is sold under the trade name Rapamune.
The immune system is vital to protect us against infectious agents (bacteria, viruses, fungi). So infection is a frequent side effect of immunosuppression in transplant recipients. Fortunately, the infections can usually be controlled by the appropriate antibiotic, antiviral drug, etc.
5% or more of transplant recipients will develop cancer within a few years of receiving their allograft. This may not seem to represent a great risk, but it does represent a 100-fold increase in risk compared to the general population. Allograft recipients are particularly prone to developing skin cancers and lymphomas.
Curiously, transplant recipients do not seem to be at any greater risk for developing the most common types of cancer in the rest of the population: cancers of the lung, breast, colon, and prostate.The chief culprit seems to be the immunosuppression that these patients have been receiving. In most cases, stopping the immunosuppression leads to regression of the cancer, but — often — rejection of the transplant as well.
The choice is usually clear for patients with allografted kidneys; they can go back on dialysis and anticipate receiving another kidney at a future date. But what of the cancer patient with a heart transplant?
Although organ transplants have helped thousands of people, much remains to be done. In particular, ways need to be found to
Xenotransplantation is the use of organs from other animals. A number of attempts have been made to use hearts, livers, and kidneys from such primates as chimpanzees and baboons — so far with limited success. One reason is that xenotransplants usually are attacked immediately by antibodies of the host resulting in hyperacute rejection. But perhaps the use of pigs as organ donors will be feasible.
However, pigs contain retroviruses (called PERV = porcine endogenous retrovirus), and there is fear that these might infect the human recipient.
Only a few transplants of pig tissue into humans have been done to date: skin grafts and grafts of pancreatic islets. A larger number of people have been temporarily hooked up to pig organs or "bioreactors" containing pig cells to provide support for their failing spleen, liver, or kidneys.
Most of these recipients have been monitored for signs of infection by PERV and — even though PERV can infect human cells growing in culture — there is no evidence that any of these people exposed to pig tissue have become infected.
As of the date of their paper, 37 PERV-inactivated piglets had been born, and several were growing normally. Perhaps pigs like these will overcome one of the hurdles to making transplantation of pig organs into humans a successful and safe procedure.
Pigs are not the only animals that contain latent viruses in their cells. Could the viruses in the cells of other kinds of animal donors infect the transplant recipient? start an epidemic?
The danger seems greater for xenotransplants from other primates. (HIV, the retrovirus that causes AIDS appears to have entered humans from a primate host — link to discussion.)
For these reasons, many biologists are urging that transplant surgeons proceed cautiously with xenotransplants.
On 7 January 2022, a 57-year-old male with heart failure received a heart taken from a genetically-modified pig.
The transplanted heart functioned adequately for 48 days. But then, for reasons that are unclear, the heart began to deteriorate and the patient died 12 days later. So although this case was a remarkable achievement, we still don't know if xenotransplantation can be made safe.
(This can cause problems. Several cases have emerged of survivors of Ebola who no longer have Ebola virus in their blood and are symptom-free but still retain live virus in such privileged sites as brain, testis, and aqueous humor of the eye where the virus has escaped attack by the immune system.)
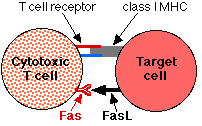
Many factors are involved in immune privilege, such as
The presence of FasL on their surface protects cells in privileged locations from immune attack because when threatened by a cytotoxic T cell, they force the T cell to commit suicide by apoptosis. Activated cytotoxic T cells express Fas on their surface. When they engage (with their T cell receptor [TCR]) a privileged cell expressing FasL, instead of killing the target, the target kills them!
So if the organs of transgenic pigs can be made to expresses human FasL, perhaps they will be resistant to T-cell mediated rejection.
The human placenta also enjoys immune privilege. It is almost as foreign to the mother as a kidney transplant from her husband would be, but unlike the kidney, she will not reject it (at least not for 9 months). In lab rats, the embryos (and the mother's endometrium) secrete corticotropin-releasing hormone (CRH). This hormone induces the expression of Fas ligand (FasL) on the cells of the placenta. Activated T cells express Fas, so any threatening T cells would commit suicide by apoptosis when they encounter FasL on their target.
| Link to more of the story of the role of Fas and FasL in apoptosis. (but note: the example you will see is the reverse of the story here; that is, the cytotoxic T cell is using its own FasL to kill a target cell that is expressing Fas but not FasL.) |
| More on apoptosis and organ transplants |
| Welcome&Next Search |