Human Embryonic Stem (ES) Cells
In other pages, I describe:
The techniques used in the early steps of each process have been achieved with human cells.
Thirteen years ago a research team led by James Thomson of the University of Wisconsin reported (in the 6 November 1998 issue of Science) that they were able to grow human embryonic stem (ES) cells in culture.
At the time of implantation, the mammalian embryo is a blastocyst.
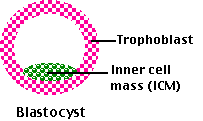 It consists of the
It consists of the
- trophoblast — a hollow sphere of cells that will go on to implant in the uterus and develop into most of the placenta.
- inner cell mass (ICM) that will develop into the
- epiblast, which will go on to form the three germ layers (ectoderm, mesosoderm, and endoderm) of the fetus as well as the extraembryonic amnion.
- hypoblast, which will go on to form the yolk sac.
The cells of the inner cell mass are considered pluripotent; that is, each is capable of producing descendants representing all of the hundreds of differentiated cell types in the newborn baby, including
Their process
- Remove the trophoblast cells from a human blastocyst (these were extras not needed for assisted reproductive technology).
- Separate the cells of the inner cell mass and culture them on a plate of "feeder" cells (mouse fibroblasts were used).
- Isolate single cells and grow them as clones.
- Test the clones.
The results
- Each successful clone maintained a normal human karyotype (unlike most cultured human cells — HeLa cells, for example).
- These cells had high levels of the enzyme telomerase, which maintains normal chromosome length and is characteristic of cells with unlimited potential to divide ("immortal").
- When injected into SCID mice, these cells formed teratomas; tumors containing a mix of differentiated human cell types, including cells characteristic of
SCID = severe combined immunodeficiency.
SCID mice lack a functioning immune system (have neither T cells nor B cells) and so cannot reject foreign tissue. (Some rare inherited diseases of humans are also called SCID. They produce a similar phenotype but involve different molecular defects. [Links]) |
Human embryonic stem cells have the potential to
- teach us about the process of human embryonic development, its genetic control, etc.
- provide a source of replacement cells to repair damaged human tissue.
As the proper signals are discovered, it will be possible to cause these cells to differentiate along a particular pathway, e.g., to form insulin-secreting beta cells of the islets of Langerhans.
Such cells might be able to replace lost or non-functioning cells in a human patient (e.g., with Type 1 diabetes mellitus).
However, there are problems that remain to be solved before this hope can be realized.
- Production of human ES cells requires the destruction of the blastocyst, and this is morally-repugnant to many people. [Link to a discussion of ways to avoid this difficulty.]
- Cell replacement therapy had better be "patient-specific"; that is, the donated cells should be genetically identical to the recipient. Otherwise, the replaced cells are at risk of being rejected by the host's immune system. [Link to a discussion of "therapeutic cloning" — a method to avoid this.
- ES cells are pluripotent and might differentiate in unwanted ways when introduced into the patient. [Link to ways to use "adult" stem cells instead.]
3 April 2021
 It consists of the
It consists of the