| Index to this page |
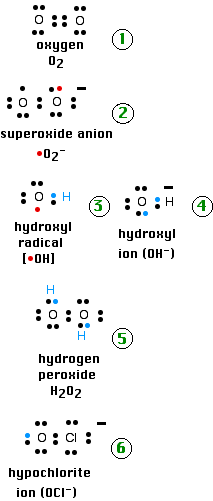 Reactive oxygen species are
Reactive oxygen species are
| Link to discussion of electron organization in atoms. |
Strong oxidants like the various ROS can damage other molecules and the cell structures of which they are a part.
Among the most important of these are the actions of free radicals on the fatty acid side chains of lipids in the various membranes of the cell, especially mitochondrial membranes (which are directly exposed to the superoxide anions produced during cellular respiration).
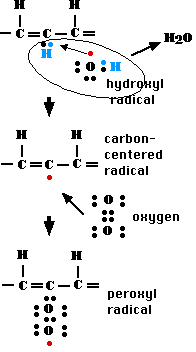
The figure shows one common series of reactions.One of the most likely (and shown here) is to react with a molecule of oxygen (O2) forming a peroxyl radical.
This might then steal a hydrogen atom from a nearby side chain making it now a radical.
One of the insidious things about free radicals is that in interacting with other molecules to gain a stable configuration of electrons, they convert that target molecule into a radical. So a chain reaction begins that will propagate until two radicals meet each other and each contributes its unpaired electron to form a covalent bond linking the two.
Two common examples:The peroxyl radical may interact with:
In both these latter cases, radical formation comes to an end but with the result that the fatty acid side chains of membrane lipids may have become so deformed as to damage the membrane.
The lipofuscin so characteristic of aging cells may be formed by these mechanisms [Link].
 These include two enzymes:
These include two enzymes:
Pharmacy shelves are filled with antioxidant preparations that people take in the hope of warding off the damaging effects (perhaps including aging) of ROS.
But it is important that the attempt to limit the production of ROS not succeed too well, because ROS have important functions to perform in the cell.
Examples:
This rare genetic disorder demonstrates the importance of ROS in protecting us from many type of bacterial infection. It is caused by a defective gene for one of the subunits of NADPH oxidase.
People with CGD have a difficult time ridding themselves of bacterial infections — especially those caused by bacteria (e.g. staphylococci, Salmonella) and fungi (e.g., Aspergillus) that produce catalase to protect themselves against the hydrogen peroxide generated by the macrophages and neutrophils that engulf them. Often the result is the development of a persisting nest of infected cells — called a granuloma.The gene for one of the subunits of NADPH most frequently mutated in CGD is on the X chromosome. Thus males are principally affected [More].
However, examination of the neutrophils of females who are carriers of the gene shows that 50% of them do not make active NADPH oxidase when they engulf pathogens. In these cells, the X chromosome with the nonmutant allele has been inactivated and converted into a Barr body. [Link to discussion]
In June 2005, two cases of successful gene therapy for CGD were reported. Blood stem cells from the patients were removed, and the active gene for the NADPH subunit inserted into them using a retroviral vector. The transformed cells were returned to the patients, took up residence in their bone marrow, proliferated successfully, and improved their symptoms.
| Welcome&Next Search |