| Index to this page |
| Some Definitions |
| Link to examples and mechanism. |
| Link to discussion of graft rejection. |
Immunological tolerance is not simply a failure to recognize an antigen; it is an active response to a particular epitope and is just as specific as an immune response.
Both B cells and T cells can be made tolerant, but it is more important to tolerize T cells than B cells because B cells cannot make antibodies to most antigens without the help of T cells.
| Discussion of T helper (Th) cells. |
T cells develop in the thymus. As they mature, recombination of gene segments creates the two chains that make up the T-cell receptor for antigen (TCR). Although the receptors on a single T cell are all alike, there is a virtually unlimited repertoire of receptor specificities created in the population of T cells within the thymus.
| Link to discussion of how T cells create receptor diversity. |
T cells whose receptors bind these epitopes so tightly that they could attack the cell displaying them are deleted by apoptosis. The T cells that survive this negative selection leave the thymus and migrate throughout the immune system (lymph nodes, spleen, etc.).
| Link to more discussion of how the T-cell repertoire is created in the thymus. |
But a question remains. The antigen-presenting cells in the thymus are certainly capable of presenting peptide fragments from the many "housekeeping" proteins found in all cells (e.g., the enzymes used in glycolysis). But there are many proteins that are expressed only in differentiated cells that are restricted to a particular tissue e.g., the insulin-producing beta cells in the islets of Langerhans in the pancreas. How is central tolerance to these proteins achieved in the thymus?
It turns out that antigen-presenting cells in the medulla of the thymus express a gene, AIRE, that encodes a transcription factor that turns on the expression of hundreds of tissue-specific genes encoding such proteins asThe AIRE protein does not seem to increase the expression of housekeeping genes. How it distinguishes between these and the tissue-specific genes to be turned on remains to be discovered. One intriguing clue is that the AIRE protein binds to chromatin whose histone H3 has no methyl groups attached to its lysine-4 ("H3K4me0". This is a mark of inactive genes. [More]
AIRE stands for autoimmune regulator. Knockout mice and those rare humans who have no functioning AIRE gene suffer from severe autoimmune disease especially of their various endocrine organs.
Thanks to the activity of AIRE, the list of the latter molecules may not be as large as we once thought.
Nonetheless, it is clear that there are mechanisms for maintaining T-cell tolerance throughout the body. What is not so clear is how many and how important each is. (The ability to demonstrate tolerance in vitro may not reflect what is important in vivo.)
Five possibilities for which there is substantial evidence:
AIRE is also active in some antigen-presenting cells in the organs of the peripheral immune system, e.g., lymph nodes and spleen. So any potentially autoreactive T cells that failed to be eliminated in the thymus can be selected against in these tissues.
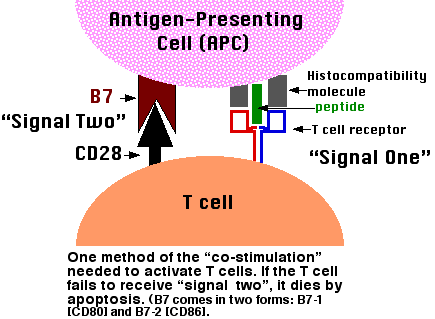 The binding of a T cell to an antigen-presenting cell (APC) is by itself not enough to activate the T cell and turn it into an effector cell: one able to, for examples,
The binding of a T cell to an antigen-presenting cell (APC) is by itself not enough to activate the T cell and turn it into an effector cell: one able to, for examples,
In order to become activated, the T cell must not only bind to the epitope (MHC-peptide) with its TCR but also receive additional signals from the APC. The receipt of these additional signals is called costimulation. One of these costimulator pairs are molecules on the APC designated B7 and their ligand on the T cell designated CD28. The binding of CD28 to B7 provides a "second" signal needed to activate the T cell.
Failure to present self antigens with sufficient costimulation leads to self-tolerance.
Examples:
A minor population of CD4+ T cells, called regulatory T cells (Treg), suppresses the activity of other T cells. They may be important players in protecting the body from attack by its other T cells.
Regulatory T cells are discussed on a separate page. Link to it.
The problem of B-cell tolerance is not so acute because B cells cannot respond to most antigens unless they receive help from T helper cells.
Nevertheless, B cells become tolerized to self components and, like T cells, this occurs both in the bone marrow (central tolerance) and elsewhere in the body (peripheral tolerance).
B cells are formed and mature in the bone marrow. In humans, over half of the developing B cells produce a BCR able to bind self components.
Any cells that produce a receptor for antigen (BCR) that would bind self components too tightly undergo a process of receptor editing. They dip again into their pool of gene segments that encode the light and heavy chains of their BCR [Discussion] and try to make a new BCR that is not a threat. If they fail, they commit suicide (apoptosis).
Despite these mechanisms, some of the B cells that migrate out of the bone marrow continue to express self-reactive BCRs and may still be able to produce anti-self antibodies. So a mechanism is needed to tolerize them out in the tissues ("peripheral tolerance").
Despite years of study, it is still uncertain what causes these diseases.
Some possibilities:
Children who inherit defective genes needed for the expression of Fas are plagued by anti-self antibodies secreted by B cells that cannot receive a FasL death signal.
These include antibodies responsible for such autoimmune disorders as:
Some autoimmune diseases may be precipitated by a prior infection. The invading pathogen may express antigens that resemble "self" (called "molecular mimicry"). These activate T and B cells. When the infection is under control, these cells may now turn against self antigens.
For example:
The human large intestine (colon) contains an enormous (~1014) population of microorganisms. (Our bodies consist of only ~1013 cells!) Most of the species live there perfectly harmlessly; that is, they are commensals. Some are actually beneficial, e.g.,
Two tolerance-inducing mechanisms have been identified.
But still unanswered is how the immune response remains capable of responding to dangerous intestinal pathogens. Perhaps these elicit the necessary "signal two".
It turns out that not only do commensal bacteria in the intestine not trigger inflammation, but their presence is needed (at least in mice) to maintain a healthy colon.
Allergists have struggled for years to find safe ways to tolerize allergic people to their allergens. This has usually involved giving a long series of injections of a special formulation of the allergen.
Examples:
On close examination, though, it appears that what the treatments are doing is shifting the immune response from the harmful, unwanted one to a harmless one (e.g., from making IgE antibodies to making IgG instead). Thus what has been induced is deviation of the immune response rather than true tolerance. (This may also be the case for transplant tolerance.)

If ways could be found to induce genuine tolerance to allografts (organs transplanted from another person), this would enable the organ to resist rejection without the need for continuous use of immunosuppressive drugs.
This photo, courtesy of the late Rupert B. Billingham (he died 16 November 2002), shows two adult white mice (strain A) that were tolerized to the cells of a black-coated strain (CBA) of mice when they were first born. Later, when adult, they were given skin grafts from the black mice. They retained these indefinitely without the need for any immunosuppression.
Although this approach is not practical for humans, it did lay the groundwork for the first successful transplants (of kidneys) in humans. It also has inspired attempts to achieve graft tolerance in humans by pretreating the recipient with blood (rich in B cells) or bone marrow of the donor.
In such cases (as well as Billingham's), it may be that tolerance of the graft is
| Organ transplantation is discussed on a separate page. Link to it. |
These are cells (both T and B) that have generated an antigen receptor (TCR for T cells, BCR for B cells) of a particular specificity, but have never encountered that antigen. There is some evidence that if they do encounter the antigen but fail to receive a "second signal", they self-destruct by apoptosis.
Both T cells and B cells that have encountered the antigen for which they are specific. If they receive a second, costimulatory signal, they become
Having received a second signal from an antigen-presenting cell, they become metabolically more active and begin rounds of mitosis (= clonal expansion).
If activated, "professional" APCs can always provide the second signal for T cells. [Link to a discussion of antigen presentation.] Dendritic cells are professional APCs, and sometimes macrophages may be.
Nonprofessional APCs can present antigen but not the second signal. B cells are nonprofessional — they present antigen to T-helper cells, but these must already be activated by an earlier encounter with professionals.
Experienced T (or B) cells that have returned to — or remained in — a quiescent state.
| Welcome&Next Search |